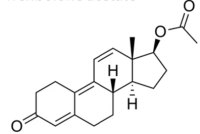
Minakem Montreal (Delmar chemicals Inc.) has been manufacturing Active Pharmaceutical Ingredients (APIs) since 1941.
It is the largest API facility in Canada, benefiting from a strong scientific and technological environment.
Located close to its U.S. customers in the New Jersey and Boston areas, Montreal also has a strong tradition of serving the entire North American market, as well as Latin America, Europe, Japan, China, and India.