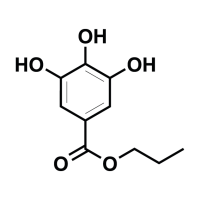
The product PROPYL GALLATE GMP is put into 2 polyethylene bags, both packed in a plastic drum/Keg.
- 20 kg → Drum 120 L Std & Milled
- 5 kg → Keg 15L Std & 20L Milled
- 1 kg (samples)→ Keg 6L Std & 10L Milled
Desiccants are put between the inner bag and outer bag
The product is stored at room temperature and dry conditions.